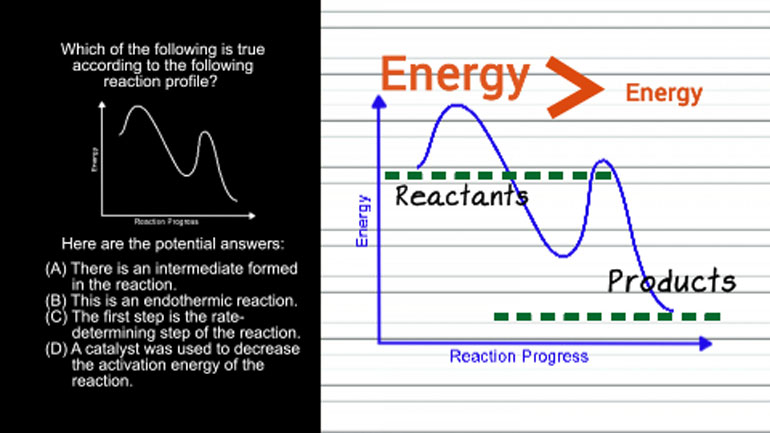
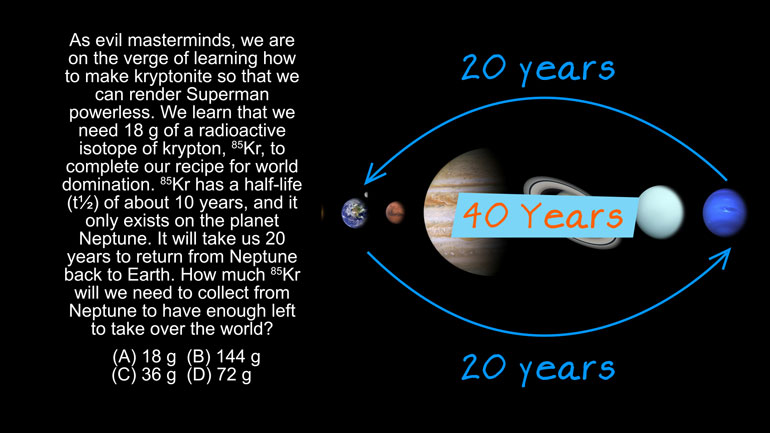
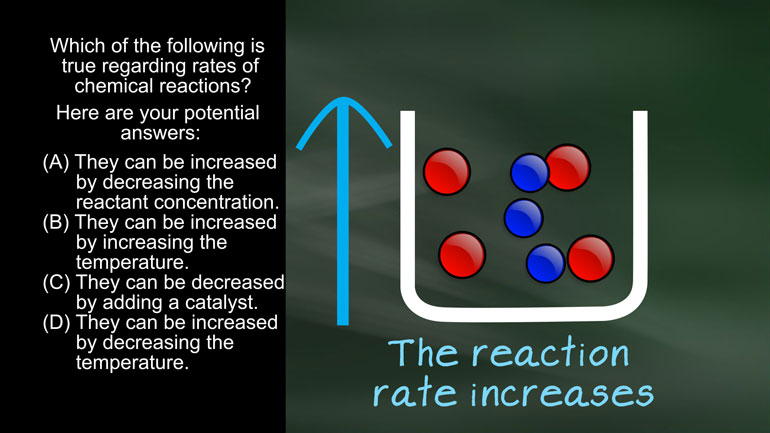
ShmoopTube
Where Monty Python meets your 10th grade teacher.
Search Thousands of Shmoop Videos
Chemical Reaction Rates Videos 15 videos
AP® Chemistry: Reaction Rates Drill 1, Problem 1. Which of the following influences the rate of a chemical reaction?
AP Chemistry 1.3 Chemical Reaction Rates. What is the overall order of the reaction?
AP Chemistry 1.4 Chemical Reaction Rates. What are the correct units for a second order rate constant?
AP Chemistry 3.4 Chemical Reaction Rates 6 Views
Share It!
Description:
AP Chemistry 3.4 Chemical Reaction Rates. What fraction of a reactant remains after four half-lives of a first order reaction?
Transcript
- 00:03
Here’s your Shmoop du jour, brought to you by glow sticks. [Glow stick bends]
- 00:07
It’s all fun and games until somebody bites into one. [Man bites glow stick]
- 00:10
Don’t do it, guys.
- 00:11
They taste terrible… according to our friend, of course.
- 00:15
We'd never be dumb enough to try that….ahem… [Man and friend with green faces]
Full Transcript
- 00:16
Here’s today’s question:
- 00:19
Raving Rodney knows that his glow sticks glow only after he cracks them.
- 00:24
What he doesn't know is that the reaction that occurs in the glow stick is called chemi-luminescence,
- 00:31
and it is a first order reaction.
- 00:33
What fraction of a reactant remains after four half-lives of a first order reaction?
- 00:41
And here are your potential answers: Raving Rodney might be a lost cause, but that [Raving Rodney appears by a woman sitting at a desk]
- 00:48
doesn’t mean you can’t learn from his mistakes.
- 00:51
Seriously, who changes their name to “Raving”…?
- 00:54
Anyway, to answer today’s question, we need to remember that the “half-life” is defined [Student highlights in a book]
- 00:59
as the amount of time it takes for something to be reduced to half its initial value.
- 01:03
Here, the molecules inside Rodney’s glow sticks that are emitting light are decreasing [Molecules in Rodney's glow stick]
- 01:08
in concentration.
- 01:09
After one half-life, the concentration of these molecules is reduced to half its initial
- 01:15
value.
- 01:16
After two half-lives, the concentration is decreased by half again, leaving Rodney with
- 01:21
1/4 of the initial concentration.
- 01:24
After three half-lives, we’re down to 1/8 the initial concentration. [Table of elapsed half lives]
- 01:26
After four half-lives, the concentration is decreased by to 1/16 of its initial value.
- 01:31
You guys get it, right? [Rodney holding a green glow stick with a green face]
- 01:34
We know you
- 01:35
You're smart.
- 01:36
You would never bite into glow sticks.
- 01:37
…Neither would we….aheeeem.
- 01:39
Anyway, our handy dandy table shows us that the correct answer is (D): only 1/16 of the
- 01:44
initial concentration of a reactant remains after four-half lives.
- 01:49
So, yeah, keep that in mind next time you crack a glowstick. [Rodney cracks a glow stick]
- 01:52
Which we know you’ll only be doing at wholesome, supervised parties, unlike Raving Rodney. [Rodney and friends at a party with glow sticks]
- 01:57
Right?
- 01:58
…Right?
Related Videos
AP Chemistry 1.3 Chemical Reaction Rates. What is the overall order of the reaction?
AP Chemistry 1.4 Chemical Reaction Rates. What are the correct units for a second order rate constant?
AP Chemistry 1.5 Chemical Reaction Rates. What is the rate law for the reaction?
AP Chemistry 3.2 Laws of Thermodynamics. What is the value for ΔG?